Forum Replies Created
-
AuthorPosts
-
Naveed RivasParticipantIn our practice we add 100ml of 20% intralipids to 250ml of normal saline. Attached are articles related to the said subject. To make it easy to find the relevant information, type ”Intralipid” in the (find) option of adobe.
You can check (Intralipid as treatment for recurrent unexplained abortion) in pubmed (under PMID: 7718096). Also there’s a trial on Clinical trial.gov with ClinicalTrials.gov Identifier: .NCT01788540.
It’s thought that it counteracts the effect of antiphospholipid antibodies which are susbected to cause repeated miscarriage.
I hope that this would be of a benefit.
https://mega.nz/file/6oxkACgZ#40y5Wl-_rm4pjKh8jlA_UBHzEtDdf7Lz3invm-6NI5c
https://mega.nz/file/f4o0RCjJ#MTgZQGWJw17HcYoNdzJEGEmqjVWKQB6babbSQ5FfmJs
Naveed RivasParticipantUsually mannitol crystalize when exposed at lower temperature especially at higher concentrations so if it is prior administration , you shall warm it to 37°C and followed by gentle agitation but if it is happened during administration so in this case you shall change the bag and you need to use always inline filter
Naveed RivasParticipantWhat s your opinion regarding k sparing diuretics like aldactone
May induce impotence & ED ?!
Decreasing dose or Switching to eplerenone ?!
Naveed RivasParticipantIn our hospital, we prepare ALTEPLASE in conc of 2mg/2ml stored in freezer to clear occluded venous catheter. Which is stable for 1 month.A regimen of alteplase is safe and effective in restoring flow to occluded central venous access devices compared to streptokinase because of the risk of life-threatening anaphylaxis.July 10, 2018 at 3:05 am in reply to: Assistance in initiating a new sterile compounding service #48528
Naveed RivasParticipantDear Rani,
The first advice to you, is to do the right thing at the beginning, otherwise you will suffer by the compromises forever..!! The right thing, in my opinion, is to have the sterile preparations as long as -out of the 24 hours- as possible.
However, here are some compromises if not 24 hours:
Some of these preparations can be done as batches, for example the TPN, and large volume parenteral solutions, you can make standardized preparations and keep them at the ward fridge. Some of the antibiotic/antifungals/IV preparations can be also done in the same way.
You can put also a laminar flow hood inside a segregated room at the hospital (as mentioned on USP 797), so you can prepare low risk preparations with BUD of 12 hours. You may let one on-call pharmacist (or more according to your production scale) to come to the hospital (if pharmacy is closed), so he/she can prepare the queue of medications which couldn’t be batched.
For STAT medications, it is not possible to cover them all if you don’t have 24 hour service. As for the USP 797, if low risk medications are prepared under non-sterile conditions, it can be used within one hour after preparing. But, again, it is still risky to standardize such a practice, and a nurse who will be the one who prepares them..!!
Keep in mind that all the above have much material and time wasting, which will raise the costs at your hospital, so I would better use this point to convince your administration to allow a 24 hour service, at least for some of the IV preparations.
I hope I helped you with my answers.
Best Regards,
Naveed RivasParticipantDownload the file as pdf in the link below..
https://mega.nz/file/r84jBSIK#IxdMpVWgQoprqgofHHf1AfuLQVsSoHWC8Yvod6vheig
Also I’ve copied the text for you. if you want a faster source.
Hope this helps.
Introduction
PN has the potential to maintain nutritional intake until the establishment of enteral feeds in preterm infants and in more mature infants with significant gastrointestinal malformation or malfunction which precludes milk feeding. PN may be the only source of nutrition, or supplied in conjunction with milk until tolerance of sufficient enteral nutrition has been achieved.
There is currently much variation within the UK in the provision of neonatal PN with regard to its use and availability as well as its constituents. The National Confidential Enquiry into Patient Outcome and Death (NCEPOD) enquiry into the care of hospital patients receiving PN in 2008 analysed data from 264 neonates in England, Wales and Northern Ireland. Only 23.5% had received PN care considered to represent good practice (1). 12 babies (4.5%) received less than satisfactory care, and room for improvement was identified in clinical care (40.5%), organisational care (9.5%) and in both clinical and organisational care (18.6%) in a majority, figures broadly similar to adult practice. A subsequent report from the Paediatric Chief Pharmacist’s Group (PCPG) endorsed concerns in the NCEPOD report regarding issues in prescribing and administration of neonatal PN, and highlighted variation in practice between units, particularly for ELBW babies (2). The PCPG defined minimum standards required to deliver PN in the NHS setting and recommended seeking consensus in best PN neonatal practice.
The ELBW infant has sufficient energy reserves to support only the first 2 – 3 days of life and even well grown term infants will rapidly become catabolic if not supplied with adequate protein and calories. All significantly preterm infants are at risk of early postnatal growth failure coinciding with the development of a cumulative nutritional deficit and linked to poorer neurodevelopmental outcome (3). Optimising growth and nutrition in the early weeks of life results in improved head and somatic growth although studies of longer-term neurocognitive outcome are lacking (4,5).
The primary purpose of neonatal PN is to achieve adequate (or as near adequate as possible) nutrition, thus permitting appropriate growth. In the absence of evidence to define appropriate growth for preterm infants, we suggest using in-utero growth as the reference. It is not clear if, when or at what rate any early postnatal fall in weight centile should be regained (6).
Administration of neonatal PN is necessarily linked with fluid balance, particularly in the first few days of life, but the nutritional needs of the neonate should be considered independently of fluid requirements. A good understanding of normal neonatal physiology is essential for those involved in prescription and administration of PN.
PN is expensive and carries significant risks including toxicity, metabolic disturbance and line-associated sepsis. All neonatal units using PN should undertake regular audit of practice, including attainment of target nutritional intake.
Who should receive PN, and when?
No study has defined the ideal criteria for administration of neonatal PN, so clinical judgement needs to be applied in balancing benefits and risks, which will vary according to postnatal and gestational age. We recommend that PN be routinely available for infants born before 30 completed weeks’ gestation (i.e. up to and including 29 weeks and 6 days), and for all infants weighing < 1250 g at birth (Table 1). Additionally, PN should normally be provided for any infant, term or preterm who has failed to establish enteral nutrition (as defined by a feed volume of > 100 mL/kg/day) by day 5 of life or who subsequently fails to tolerate enteral nutrition over a significant period. Infants of any gestation who are not predicted to establish enteral nutrition by day 5 should also be considered for PN. All services caring for neonates meeting these criteria should have 24-hour access to PN, 7 days a week.
Table 1: Indications for PN in neonates
Absolute Indications
Gestational age < 30 completed weeks (i.e. up to and
including 29 weeks and 6 days)
Birth weight < 1250 g
Failure to establish enteral nutrition* by day 5 of life,
regardless of gestation or birth weight
Inability to tolerate enteral nutrition for a period likely to result
in a significant nutritional deficit**
*Enteral nutrition defined as > 100mL/kg/day milk
**The likelihood of a significant nutritional deficit depends on
many factors including age, gestation, and birth weight. PN
should be considered for any baby > 5 days of age who
becomes unable to tolerate enteral feeds for > 24 hours
Relative Indication
Any baby > 30 weeks gestation considered unlikely to
establish enteral nutrition by day 5.
The duration of PN, as well as the period of intolerance of enteral feeding considered significant will be determined by gestation, birth weight and other morbidities as well as local preferences for advancement of enteral feeds. In some cases secure venous access may also be a consideration. Local nutrition guidelines should be agreed and implemented, as this helps to achieve consistency of practice and shortens time to full enteral feeds (7). It is important that nutrition guidelines are regularly reviewed as the results of feeding trials become available.
Newborn infants meeting an absolute criterion for PN should be started on intravenous (IV) glucose and amino acid (AA) solution as soon after birth as possible and IV lipid added within 24 hours. Normally PN will be commenced immediately after a central venous line has been sited and its position deemed acceptable although PN may be administered peripherally (see below). Similarly, there should be no delay in commencing PN in older, more mature babies once a decision for PN has been made. We strongly recommend that nutritional intake be considered separately from fluid management, but acknowledge that for relatively stable babies a stepwise increase in total volume of infused fluid of a constant glucose and AA solution may be considered the most appropriate way to manage PN in the first few days of life.
Constitution of PN (summarised in Table 2, appendix)
Water and electrolytes:
Although the primary purpose of neonatal PN is to achieve adequate nutrition, fluid and electrolyte balance will need to be considered, particularly in the first few days of life when there may be rapid changes in both intracellular and extracellular body fluid compartments. It is reasonable to start on day 1 with fluid volumes of 60 – 100 mL/kg/day, increased according to the baby’s clinical condition. Measured fluid balance is unreliable and must be interpreted with caution; daily weighing of the smallest and sickest babies is essential. To facilitate consistency of nutrition, PN content may be calculated to provide daily nutrient requirements in a minimum acceptable fluid volume, with additional fluids and electrolytes infused separately as required. Such concentrated PN solutions allow prompt changes in fluid and electrolyte management without need for changes to the PN solution but require more complex calculation of fluid and electrolyte intake, with increased potential for prescription and administration error. For a majority of stable babies, fluid requirements are reasonably predictable and can be managed by stepwise increase in volume of standard PN solutions, recognising that this practice is unlikely to meet full nutritional requirements in the first week of life.
1. Sodium
Following the immediate postnatal physiological diuresis, at least 1 – 3 mmol/kg/day of sodium should be provided in PN from 48 – 72 hours of life. Faecal and renal losses in the smallest preterm infants may result in much higher sodium requirements. Small amounts of sodium may safely be given in the first two days, and will necessarily accompany phosphate supplementation of PN fluids. More evidence is required to define sodium requirements for optimal early nutrition. Measuring urinary sodium losses may help to guide sodium supplementation after the first few days.
2. Potassium
Extracellular potassium concentration is affected by blood pH and may not reflect intracellular concentration. Assuming adequate urine production, 1 – 2 mmol/kg/day potassium should be commenced from 48 – 72 hours of age, increased to 2 – 3 mmol/kg/day (or more as required) from week two. Requirements may be higher in preterm infants and will be affected by the presence of renal or gut pathology, external cerebrospinal fluid drainage or diuretic therapy. Small amounts of potassium will be present in PN fluids containing acetate salts.
3. Chloride
The intake and output of chloride usually parallels that of sodium, but chloride losses will be modified by bicarbonate status. Additional chloride is often infused with other electrolytes (e.g. potassium chloride); since excess chloride administration may result in metabolic acidosis, use of acetate salts should be considered (8). Recommendations for practice
- Regular monitoring of water and electrolyte balance in neonates receiving PN should be undertaken. The best guide to overall fluid balance is measurement of body weight; electrolyte balance should be considered in conjunction with plasma electrolytes. ELBW babies will normally require 12 hourly monitoring, at least for the first 48 – 72 hours of life.
- Only small amounts of sodium are generally required in the first 48 hours of life.
- Refinements to fluid and electrolyte balance may be achieved either by changes to the total prescribed volume of PN or by titration of additional fluid and electrolyte solutions in addition to concentrated PN solutions. Whichever method is practised, nutrient provision should be considered in addition to fluid balance.
Energy:
PN must supply sufficient energy to cover energy expenditure and losses in addition to growth. Resting energy expenditure increases over the first few weeks of life, with significant inter-individual variability (9); in a relatively stable small preterm infant towards the end of the first week resting energy expenditure will be around 80 – 95 kcal/kg/day. Fetal energy accretion is approximately 24 kcal/kg/day, so provision of 100 – 120 kcal/kg/day is recommended for neonatal PN (10). Energy requirements are slightly less in the parenterally, compared to enterally fed infant. Since protein is required for tissue repair and new tissue deposition, the majority of energy provision in PN should be non-nitrogen (i.e from carbohydrate and/or lipid).
Recommendation for practice
- For most stable neonates PN should provide a total of 100 – 120 kcal/kg/day (including 85 – 100 non-nitrogen kcal/kg/day) by 72 hours of age. Carbohydrate: Glucose is the major source of energy for most metabolic processes in the body and is also an important source of carbon for the synthesis of non-essential fatty and amino acids. D-glucose (dextrose), the main carbohydrate and energy source in neonatal PN, provides 3.4 kcal/g. It should be noted that carbohydrate provides on average 4 kcal/g; this latter formula is commonly used for calculation of PN content.
Considerations
1. Glucose production and utilisation
Term infants produce around 5 mg/kg/min glucose. Preterm infants can produce up to 5 mg/kg/min glucose from glycogenolysis and a further 4 – 5 mg/kg/min from gluconeogenesis. This production continues in the face of IV infusion of adequate amounts of glucose, and may contribute to hyperglycaemia. Glucose utilisation in term infants is around 3.5 – 4.5 mg/kg/min but may be double this in preterm infants (10).
2. Provision of glucose in PN
ESPGHAN guidelines advise daily glucose intakes of 5.8 – 17.3 g/kg (4 – 12 mg/kg/min) in the neonatal period (11). PN protocols providing energy at the upper end of this recommendation in conjunction with higher protein are associated with more rapid return to birth weight and improvement in both weight gain and head circumferential growth, without increase in relative body fat (5,12,13).
3. Risks of excessive glucose intake
- Excess fat deposition
Abnormal body composition at term equivalent age, with excess fat and a lack of lean mass has been described in preterm infants (14). This may be due at least in part to a combination of excessive energy provision and inadequate provision of protein, and is likely to be modifiable depending on early nutrition strategy (15).
- Hyperglycaemia
Neonatal hyperglycaemia is reasonably consistently defined as a whole blood glucose concentration > 6.9 mmol/L or a plasma or serum glucose concentration > 8.3 mmol/L. Hyperglycaemia is common in very preterm infants, but although associated with an increased risk of mortality and many other complications of prematurity there is no evidence of a causal relationship (16).
4. Management of neonatal hyperglycaemia in association with PN
Hyperglycaemia may be tolerated providing that no metabolic complications such as dehydration and metabolic acidosis arise. Treatment options include glucose reduction and/or insulin therapy. There is no definitive evidence to guide practice (16).
- Glucose reduction
Glucose reduction results in lower energy intake with potentially impaired utilisation of AA and a risk of growth failure. Lower energy intakes in the first week of life are associated with poorer neurodevelopmental outcome (17). Glucose provision should not be reduced below 5.8 g/kg/day (4 mg/kg/min).
- Insulin therapy
The main risk relating to insulin therapy is hypoglycaemia; this risk is likely to be less if insulin is used to treat, rather than prevent hyperglycaemia (18). Nutritional strategies promoting rapid early growth may have implications for the later development of metabolic syndrome (19) although there is currently no evidence linking the latter with insulin therapy in preterm infants. Studies demonstrating improved neonatal growth with high protein and energy intakes have used insulin therapy to treat hyperglycaemia (5,12,20).
- AA and lipid intake
AAs stimulate insulin secretion in preterm infants and early AA administration is associated with a lower incidence of hyperglycaemia (21). Increasing early glucose and AA intake simultaneously does not increase the risk of hyperglycaemia. There is some evidence that IV lipid may exacerbate hyperglycaemia via stimulation of gluconeogenesis (18).
Recommendations for practice
- Regardless of gestation, the minimum amount of glucose that should be provided in the first 24 hours is 5.8g/kg/day.
- For both preterm and term infants, glucose intake should be increased as tolerated. The maximum intake should not exceed 17.3 g/kg/day.
- A local policy should be agreed for the management of hyperglycaemia. When interventions are deemed necessary, these may include reduction of glucose intake, use of insulin, early AA administration and/or avoidance of high early lipid provision.
Protein/amino acids:
Protein is required for cell structure and function and is provided in PN in the form of AA. PN protein is estimated from the nitrogen content of the fluid; depending on the AA formulation, protein content will be up to 12% lower than the AA content. Calculating PN protein intake (g/kg/day) may be useful in order to allow total (enteral and parenteral) protein intake to be ascertained. Neonatal PN AA formulations are designed to provide a full range of AA including the essential, conditionally essential and non-essential AA required for protein synthesis, and to reflect as closely as possible umbilical cord blood or human milk protein AA profiles. Formulations are limited by solubility and stability of AA; in addition, the bioavailability of some AA is affected by metabolic demands other than protein synthesis and by metabolism of milk proteins in the gut (22). Since the intestine is bypassed in PN, the AA requirement should be less than for enterally fed babies, but this may be partially offset by unpredictable absorption and metabolism of specific amino acids.
Considerations
1. Protein utilisation and accretion
The minimum protein intake required to prevent loss of existing tissue protein is 1.5 g/kg/day (11). In infants of less than 29 weeks’ gestation, starting 1.5 – 2.4 g/kg/day parenteral protein immediately after birth achieves a positive nitrogen balance in the first 48 hours of life (23). Increasing parenteral protein intake thereafter up to 3.6 g/kg/day in association with 90 kcal/kg/day of non-protein energy further improves nitrogen retention without significant biochemical derangement other than increased plasma urea concentration (24,25). There is a paucity of evidence in more mature preterm infants and in term infants.
2. Provision of AA in PN
Interpretation and comparison of published studies is difficult due to variation in study populations and differences in rates of incrementation of protein and calories, energy:protein ratio and outcome measures.
- Starting dose
Nitrogen balance is not improved by starting doses of > 3.5 g protein/kg/day compared to starting doses of 1.5 – 2.4 g/kg/day (24,26).
- Target protein and energy intakes
Protein intakes of 3.5 – 4 g/kg/day in association with non-protein energy:protein ratios of 21 – 25 are associated with improved growth outcomes (4,12,20). Similar protein intakes in association with lesser non-protein energy:protein ratios do not however improve nitrogen retention or lean body mass compared to protein intakes of 2.7 g/kg/day (15,24). The ratio of energy:protein in PN almost certainly influences utilisation of AA and promotion of optimal body composition and although there is a paucity of evidence, it is likely that 18 – 25 non-nitrogen kcal are required per g protein (10,15). Attainment of target protein dose may be challenging, particularly in the first two or three days and attention needs to be paid to the actual protein intake achieved. The actual daily intake should reach the target daily intake before day 5 in order that the total target protein intake for the first week of life can be met. There is evidence that using concentrated PN solutions results in better attainment of target protein intake (20).
- Optimal neonatal PN AA formulation
There are no studies comparing different neonatal PN AA formulations. Current formulations do not result in optimal plasma AA profiles, which may have implications for the efficiency of protein synthesis and carry a risk of toxicity (27,28). Many of the AA products in current use have formulations that are 25 years old and safety data are largely based on small historical preterm populations. There is a need for new evidence.
3. Longer term outcomes
Observational studies have associated higher early protein and energy intake with improved neurodevelopmental outcome at 18 months but data from randomised controlled studies are limited. The SCAMP study showed improved early head growth with increased protein and energy intake in very preterm infants (4) but in the similarly sized NEON study increased amino acid provision without increased energy intake was associated with poorer head growth (15). There has been no randomised controlled trial of increased parenteral protein and/or energy powered to evaluate neurodevelopment or longer term metabolic complications as a primary outcome (29).
Recommendations for practice
- Preterm infants
- Preterm infants meeting at least one criterion for PN at birth should be started on protein as soon as possible at a dose of 2 – 2.5 g/kg/day.
- Target parenteral protein intake should be 2.7 – 4 g/kg/day by day 5 of life, regardless of age at commencing PN and assuming adequate calorie intake.
- Term and larger preterm infants (> 34 weeks’ gestation unless significant intrauterine growth restriction)
- Target protein intake should be 3 g/kg/day by day 5 of life.
- The choice of parenteral AA formulation for each network provider should involve dietetic as well as pharmacy expertise.
Lipid:
As well as fat and calories for growth, IV lipids provide n-6 and n-3 essential fatty acids for brain development; without supplementation ELBW infants risk becoming deficient in essential fatty acids within two days of birth (30). IV lipid reduces lipogenesis and is associated with decreased energy expenditure, reduced oxygen consumption and carbon dioxide production and improved nitrogen retention. 20% lipid emulsion provides 2 kcal/mL, more than five times the calorie density of 10% glucose and AA solutions.
Considerations
1. Lipid utilisation and accretion
The fetal requirement for fatty acids increases during the third trimester from approximately 1.0 g/kg/day to just over 2.0 g/kg/day at term (10).
2. Provision of lipid in PN
a. Starting dose
IV lipid is generally well tolerated from birth even in ELBW babies (31,32). VLBW babies given 2 g lipid/kg/day on day one and 3 g /kg/day on day two have improved nitrogen retention compared to VLBW babies given glucose and AA solution alone (24).
b. Target lipid intake
IV lipid provision of up to 3.5 g/kg/day in the first week of life is well tolerated in VLBW infants and associated with improved energy intake and weight at hospital discharge (32).
c. Which lipid preparation to use?
Newer lipid formulations include medium chain triglycerides, olive oil and fish oils as well as soybean oil; these may beneficial in babies with liver disease but long term data are lacking (33). A weak association between these newer lipids and reduction in sepsis has been described but there is no clear evidence of benefit or harm from routine use in very low birth weight babies (15,24,34). 20% lipid emulsion is lower in phospholipids than 10% emulsion and thus likely to result in better plasma clearance of trigycerides (35).
3. Tolerance and risks of IV lipid
IV lipid tolerance is dependent on many factors, including gestation, rate of infusion, the metabolic and growth status of the baby, and the preparation of lipid. Infusion of lipid over 24 hours is better tolerated than intermittent infusion. Tolerance of IV lipid is usually assessed by measuring plasma triglycerides but there is little evidence to define normal plasma triglyceride concentrations in the newborn. Hyperglycaemia may be exacerbated by higher rates of lipid infusion, particularly in the smaller babies (24,36) but historical concerns regarding IV lipid and hyperbilirubinaemia, chronic lung disease, impaired immune function, sepsis, altered platelet function, NEC, and ROP have not been proven (31). Any potential risks of IV lipid are outweighed by the benefits of early provision of essential fatty acids, particularly for the smallest babies
Recommendations for practice
- IV lipid should be given in addition to aqueous PN in a dose of 2 g/kg/day on the first day of life and increased daily to a maximum of 3.5 – 4 g/kg,depending on lipid and glucose tolerance. It is noted that some newer lipid formulations are licensed only to a dose of 3 g/kg/day.
- A 20% lipid emulsion is the preparation of choice; this should be continuously infused over 24 hours.
Micronutrients:
1. Calcium, phosphate and magnesium
Calcium (Ca), phosphate and magnesium (Mg) are required for bone structure and both muscle and nerve function. Ca should be provided in PN from day 1 to avoid early hypocalcaemia secondary to delayed secretion of parathyroid hormone. Organic phosphate compounds should be used in PN to achieve bone mineral accretion comparable to that in utero (11).
Phosphate requirements are likely to be higher when protein intake has been maximised and growth optimised; phosphate and Ca should be provided to preterm infants initially in a 1:1 molar ratio (26,37); subsequently higher doses of phosphate may be required. A reasonable initial parenteral dose for all neonates would be 1.5 mmol/kg/day each of Ca and phosphate and 0.18 – 0.2 mmol/kg/day Mg.
2. Trace elements
Although nutritionally essential, precise requirements for trace elements in the premature neonate remain uncertain. Commercially available additions can only be added to aqueous PN, although stable all-in-one neonatal PN solutions may become available in the future (38). The addition of trace elements adversely affects the long- term stability of standard PN bags and so it is reasonable to commence standard, unsupplemented PN solutions out of hours, with trace elements added to the first routine PN fluid change.
While most trace elements appear to be safe, IV iron carries a risk of oxidant injury and is not recommended, at least for the first three weeks. Most neonates can be managed with enteral iron supplements once PN has been discontinued Further information on trace elements is summarised in Table 2, appendix.
3. Vitamins
Preterm infants are born with reduced stores of fat soluble vitamins and little, if any, stores of water soluble vitamins. Current guidelines for vitamin supplementation of preterm infants are based largely on expert opinion, with most data obtained from small historical studies of moderately preterm babies (11,39). In practice, IV administration of fat and water soluble vitamins is limited by the availability of suitable multi-vitamin preparations.
Vitamin supplements must be added to PN fluids in a pharmacy aseptic unit. Ready mixed lipid and vitamin preparations are now available; a decision to use such products will need to consider convenience, safety and cost.
In the absence of evidence of efficacy or toxicity, it is recommended that both a fat and a water soluble multivitamin preparation be added to PN fluids at the earliest practical opportunity.
Recommendations for practice
- PN should be commenced with phosphate and Ca in a molar ratio of 1:1, adjusted as required to maintain normal plasma Ca and phosphate. It is likely
that higher amounts of phosphate will be required for the smallest preterm infants.
- Adjustment of Ca and phosphate levels in PN solutions should be undertaken with caution as there is potential for precipitation of salts. More soluble organic phosphate salts should be used.
- Neonatal PN should be supplemented with trace elements using a commercially available preparation in the manufacturer’s recommended dose as soon as practical after commencing PN.
- Both fat and water soluble vitamins should be given in the manufacturer’s recommended doses within 48 hours of commencing PN.
- Iron should not be added to PN fluids except in those infants wholly dependent on PN after 3 weeks of age. When iron has been added to PN, serum ferritin should be monitored.
Practical considerations
Following a decision to start PN, consideration must be given to prescription, preparation and administration. Safe execution of each of these steps requires a multidisciplinary team including doctors, dietitians, pharmacists and other members of the pharmacy team and nursing staff. Every stage of the process of providing PN to a baby carries risks, all of which are discussed in the PCPG report (2). All neonatal units providing PN should have access to pharmacy support, at least during regular working hours, and protocols should be in place to meet the recommendations of the PCPG.
Prescription of PN:
Initiation of PN in the neonatal unit (NNU) should follow locally agreed protocols. A decision to initiate, or not to initiate, neonatal PN out with agreed criteria should only be made by an experienced healthcare professional, usually the senior attending clinician. As there is significant potential for error in prescription of PN, this should only be undertaken by suitably trained persons in accordance with locally agreed guidelines. Prescriptions should be reviewed daily. Ideally prescription of PN will involve a specialist neonatal pharmacist and a specialist neonatal dietitian as well as the clinician, but it is recognised that this may not be achievable in all NNUs. In compiling local guidelines separate consideration should be given to the nutritional and fluid requirements of the neonate, particularly in the first week of life, and both specialist pharmacist and dietetic input must have been sought. When concentrated PN solutions are chosen, guidelines must include clear and comprehensive instructions for manipulation of additional fluids. PN and total fluids should be prescribed based on birth weight until this has been exceeded; thereafter the baby’s greatest recent weight should be used unless there is significant oedema. Standardised prescription charts reduce the likelihood of error, and electronic prescribing eliminates the risk of transcription error. Standardised request forms for PN help to ensure a controlled, validated system of documentation of prescribing. Any change in infusion rate from that on the label of the PN solution must be within the maximal permitted rate of infusion for the solution, and must be clearly documented on the patient’s PN prescription chart. The clinician who is accountable for deciding upon the nutritional needs of the baby and the formulation of PN to be administered should also sign the prescription.
Compounding of PN:
The complexities of compounding of PN should not be underestimated.
1. Preparation and storage
PN must always be prepared in a pharmacy aseptic unit. PN solutions should be stored in a fridge when not in use, and allowed to reach room temperature before administration. This allows the solution to de-gas and reduces the likelihood of bubble formation. To reduce the possibility of contamination and/or problems with incompatibility of additives and instability of solutions, changes must not be made to PN formulations at ward level. Urgently required alterations to glucose or electrolyte provision can be achieved without manipulating PN solutions.
2. Additions to PN solutions
Drugs should not be added to PN solutions. The common practice of adding heparin to PN affects lipid bonds and has not been shown to increase catheter patency so is not recommended (40). Additions to PN solutions must not be made outside the pharmacy aseptic unit.
3. Standardised versus bespoke PN solutions
Standardised PN solutions are associated with lower risk of compounding and prescribing error, and will be suitable for a vast majority of neonatal patients. It is recommended that NNUs and networks use standardised PN bags whenever possible, and that a decision to use a bespoke PN solution is only made by a senior clinician, in conjunction with a specialist neonatal pharmacist and ideally a specialist neonatal dietitian. It is recognised that specialist dietetic support is not available in all NNU. Suitable PN can almost always be provided for the first 24 – 72 hours by having 24 hour access to standardised PN solutions. Several standardised formulations of neonatal PN are currently available in the UK, including some concentrated formulae; the composition of locally agreed PN solutions should be regularly reviewed and reflect the results of properly conducted randomised clinical trials with relevant safety data. Experience exists with glucose contents ranging from 7.2 to 16.7 g/100 mL and protein ranging from 2.0 to 3.8 g/100 mL (2.3 – 4.3 g AA/100 ml).
Administration of PN:
PN differs from simple glucose and electrolyte IV fluids in many regards including pH and tonicity. The risks of extravasation injury and contamination are increased with PN, and there is a need for enhanced biochemical monitoring. PN is an independent risk factor for neonatal sepsis and catheter-associated bloodstream infection (41). Cost is also an important factor, added to by dedicated line sets and specialised filters. Wastage should be kept to a minimum.
1. Peripheral versus central lines
Ideally PN should be administered via a central line, particularly if the patient is expected to be PN dependent for more than a few days. In neonates this requires either a properly sited umbilical venous catheter or a peripheral percutaneous IV central catheter (PICC). Careful attention must be paid to maintaining sterility of central lines both during and after insertion, and local protocols for line care should be in place. Ideally a designated central line should be used exclusively for PN and not accessed for blood sampling but it is recognised that this is not always practical in the NNU setting. Local policies should be agreed with regard to insertion and management of central lines, and all staff trained accordingly. The reader is referred to BAPM guidance on use of central venous catheters in the newborn (42). If other infusions are to run alongside PN then compatibility must be confirmed (11). Central venous line position must be confirmed by X-ray before use (43). The preferred position for the central line tip is either the inferior vena cava (ideally at level of the diaphragm) or the superior vena cava (long-line only) (44), but it may not be possible to insert PICC lines to this level. Atrial positioning should be avoided due to risk of atrial perforation and cardiac tamponade (45). Even when the position of a central line has been confirmed by X-ray, practitioners need to be aware of the possibility of line migration. Some NNUs will choose to administer PN via peripheral venous cannulae, often (but not exclusively) as a short-term measure until central access can be achieved or to maintain the delivery of nutrition while treating a central line-related infection. When PN is administered via peripheral cannulae the risk of extravasation injury needs to be balanced against the risks or difficulties associated with obtaining/maintaining central venous access, and the nutritional needs of the infant. There is a lack of consensus around the maximum osmolarity of IV fluids which can safely be administered via peripheral cannulae; recommendations based on both paediatric and adult data vary from 600 – 900 mOsm/L. There is a dearth of neonatal studies. Osmolarities of 900 – 1000 mOsm/L are not clearly associated with higher risk of line-related events although redness or minor swelling at the peripheral site are common; only one significant event (marked swelling, blanching of area, skin necrosis or blistering and decreased or absent pulses below the site) was reported in a total of 668 days of peripheral PN (46). Osmolarities greater than 1000 mOsm/L have been associated with increased complications including thrombophlebitis and infiltration (47). Infusion of lipid via the same peripheral catheter as the aqueous component may have a vascular protective effect (48). PN that is not suitable for peripheral line administration must be clearly labelled: ”to be given by central line only”; this is likely to include most concentrated PN solutions (2). The osmolarity of 12.5% dextrose is 630 mOsm/L.
2. Commencement of PN infusion
Priming of lines and giving sets should be done under full aseptic conditions. A final safety check at the point of administration of PN should be performed, including checking the labels of PN solutions against the request form for: name of patient and identifying number, route of administration (central or peripheral), date for infusion, expiry date and appearance of the PN solution(s) (2). NNUs/networks should have agreed protocols, and ensure that staff members are appropriately trained in the commencement of PN infusions
3 Filters, giving sets and infusion times
The use of in-line filters to reduce the risk of contamination of PN fluids by bacteria, endotoxins and particulates is widely practised and currently recommended by ESPGHAN. Early data in adults suggested efficacy of in-line filters, but the practice is increasingly being questioned, with no evidence of benefit in some recent studies in both adults and children. There is a paucity of evidence in neonatal practice (49). There are two main IV filter pore sizes; the 0.22 micron filter used for aqueous solutions, reported to remove air, microorganisms and particulate matter as well as endotoxins released by gram-negative bacteria (50) and the 1.2 micron filter used for larger molecule solutions including lipid. Based on data from two studies including 530 neonatal patients, use of in-line filters in central line giving sets changed every 96 hours does not reduce mortality or the incidence of proven or suspected septicaemia compared to unfiltered lines changed daily. In-line filters are cost effective when IV fluids and giving sets are changed 96 hourly compared to 24 hourly for unfiltered lines (51). Changing PN bags 48 instead of 24 hourly offers considerable cost savings and does not result in increased central line-associated blood stream infection in term and larger preterm infants (52). Extending infusion time for glucose and AA solution to 96 hours in association with in-line filter use may be safe but this practice has not been directly compared with shorter infusion times using the same filters. There is some evidence to suggest that it may be acceptable to use lipid giving sets for 48 hours but rates of infection increase with 72-hourly lipid giving set changes compared to 24 hourly. A pragmatic and safe approach would be to change lipid syringes and giving sets every 24 hours (52,53).
4. Shielding from light
Light exposure is associated with peroxide generation in PN solutions which creates oxidant stress, increases degradation of vitamins and has been implicated in both impaired lipid tolerance and slower advancement of enteral nutrition (54,55). In a recent randomised controlled study, protection of PN solutions from light was not associated with reduction in either bronchopulmonary dysplasia or death (56). Peroxide has bacteriostatic properties and so shielding PN solutions from light might conceivably enhance the risk of late-onset sepsis but this has not been proven. On balance, we recommend that all PN bags and giving sets should be protected from
light.
5. Vitamin administration
Both fat and water soluble vitamins should be given with the lipid emulsion to improve vitamin stability and protect lipid preparations from peroxidation (54). Water soluble vitamins may be added to aqueous PN if lipid is not being given.
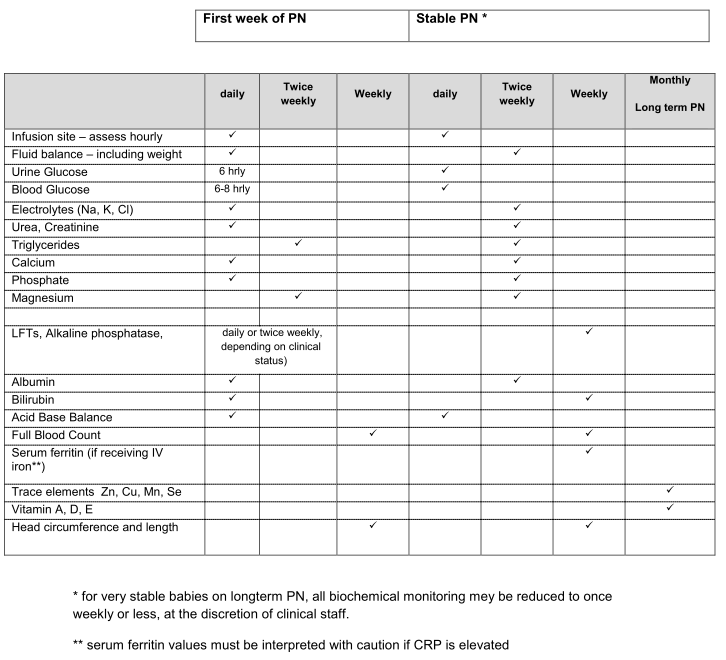
6. Monitoring
Unexpected biochemical instability as a consequence of PN is rare but may have serious consequences. Routine biochemical monitoring is generally accepted as best practice and should take into consideration the duration of PN, infant gestation, co- morbidities and other administered medicines (2). The individual responsible for reviewing biochemical results and taking appropriate action when abnormal values are observed must be clearly identified on a daily basis. We have included a suggested schedule of monitoring based on ESPGHAN recommendations (11) which should be implemented according to the baby’s clinical condition (Table 3, appendix). Tolerance of IV lipid is usually assessed by measuring plasma triglycerides but there is little evidence to define normal plasma triglyceride concentrations in the newborn. Plasma triglyceride concentration > 3 mmol/L is common regardless of the amount of lipid provision (24,32) and so it is reasonable to accept values of up to 3 mmol/L as within normal. Unwell, septic infants are more likely to demonstrate poor lipid tolerance.
Low selenium status has been implicated in oxidative diseases such as bronchopulmonary dysplasia and retinopathy of prematurity, but there is insufficient evidence to recommend routine monitoring of plasma selenium levels (57). There is no need to monitor trace element status routinely for the first 3 weeks of PN treatment but for infants receiving long-term PN (defined as > 3 weeks with minimal or no enteral feeds) additional monitoring on a monthly basis for trace element status (including copper, manganese, selenium and zinc) and fat soluble vitamin status should be considered.
Recommendations for practice
- All NNUs/networks should have agreed protocols for the initiation of neonatal PN. Variation from protocol should only be made by a suitably experienced healthcare professional.
- Neonatal networks should aim to implement standardised, electronic PN prescription charts.
- Additions to PN fluids should only be made in a pharmacy aseptic unit.
- Standardised PN solutions should be used whenever possible.
- PN should ideally be infused via a central line; the position of which must be confirmed by X-ray. PN formulations with an osmolarity greater than 1000 mOsm/L should not be administered peripherally.
- Commencement of PN infusions must be done under aseptic conditions, and with locally agreed checks in place to minimise error.
- Aqueous lines used with 0.22 micron in-line filters may safely be left undisturbed for up to 48 hours; lipid lines should be changed at intervals of 24 hours.
- Fat (and ideally water) soluble vitamins should be added to lipid emulsion, to improve vitamin stability.
- Regular biochemical monitoring of neonatal PN patients should be undertaken, according to locally agreed protocol.
Weaning PN
Appropriate minimal enteral feeds, preferably maternal or donor breast milk should be given in conjunction with PN wherever possible to prevent gut atrophy and encourage gut adaptation, as well as reduce the risk of PN-associated liver disease. Weaning of PN should be considered once the baby is stable and able to tolerate some enteral feed. Weaning of PN should be described in local feeding guidelines and both aqueous and lipid phases reduced proportionally as enteral feeds increase. The rate of weaning will depend upon the baby’s clinical condition and may be limited by the availability of maternal expressed breast milk. Care needs to be taken to ensure that electrolyte requirements are met as the volume of aqueous PN is decreased; sodium may need to be added to milk or given as an oral supplement. Occasionally PN will need to be continued together with milk feeds for prolonged periods if milk tolerance is impaired. PN should generally be continued until at least 75% of nutritional requirement is tolerated enterally (58). For preterm babies after the first few days of life, this would generally be a milk intake of at least 120ml/kg/day.
Recommendations for practice
- Weaning of PN should be undertaken according to locally agreed protocol which ensures preservation of protein and energy intakes during the transition phase. The policy should include a strategy for proportional reduction of all macronutrients.
- PN should not normally be ceased until at least 75% of total daily nutritional requirements are provided via enteral feeds.
Audit of practice
All NNUs administering PN should have a programme for regular audit of practice. Networks and individual NNUs should be aware of their compliance with local guidelines for administration of PN as well as incidence of PN-associated morbidities, including sepsis and catheter complications. Equally importantly, the efficiency of PN in delivering target nutrition should be regularly reviewed since actual provision of macronutrients may be considerably different from that prescribed and/or intended (58).
Conclusions:
Neonatal PN is an essential element of modern neonatal care with potential to influence both short and longer term outcomes. All neonatal patients in the UK deserve equal access to PN, prescribed and administered according to carefully considered local guidelines. This Framework for Practice outlines how this may be achieved.
Table 2: Recommended macro and micro nutrient intake for neonatal PN (11).
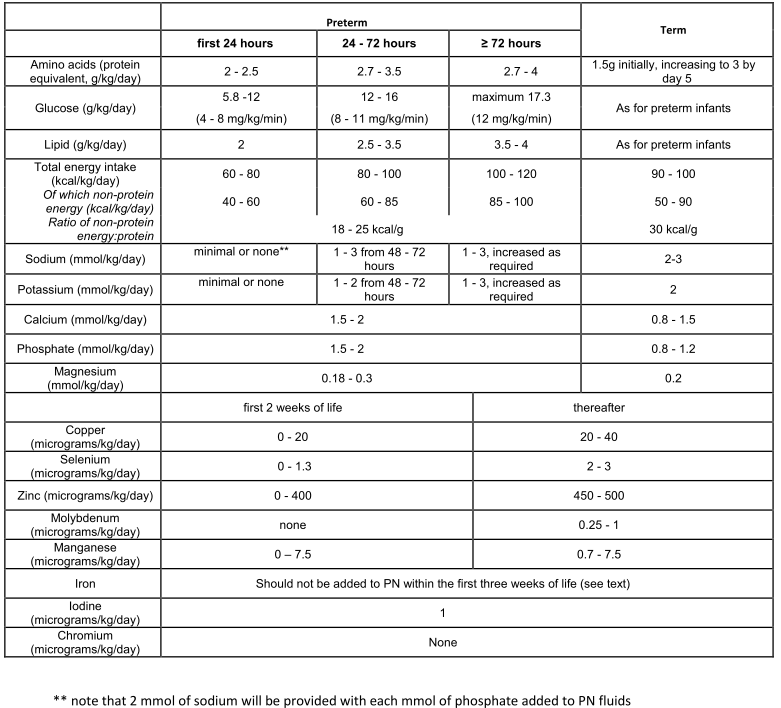
Table 3: Suggested monitoring of neonates receiving PN.
References:
- Stewart JAD, Mason DG, Smith N, Protopapa K, Mason N, on behalf of CEPOD. A mixed bag; an enquiry into the care of hospital patients receiving parenteral. 2010 nutrition. 2010. available at http://www.ncepod.org.uk/2010report1/downloads/PN summary.pdf.1a. Accessed 3/5/15
- Improving practice and reducing risk in the provision of parenteral nutrition for neonates and children: Report of the Paediatric Chief Pharmacists Group, November 2011. available at http://www.rpharms.com/support-pdfs/minimising- risk-pn-children-%286%29.pdf. Accessed 8/5/15
- Wood NS, Costeloe K, Gibson AT, Hennessy EM, Marlow N, Wilkinson AR. The EPICure study: growth and associated problems in children born at 25 weeks of gestational age or less. Arch Dis Child Fetal Neonatal Ed 2003;88;F492-500
- Morgan C, McGowan P, Herwitker S, Hart AE, Turner MA. Postnatal head growth in preterm infants: a randomised controlled parenteral nutrition study. Pediatr 2014;133:e120-8.
- Blanco CL. Gong AK Schoolfield J, Green BK, Daniels W, Liechty EA, Ramamurthy R. Impact of early and high amino acid supplementation on ELBW infants at 2 years. J Pediatr Gastroenterol Nutr 2012;54:601-7
- Cole TJ, Statnikov Y, Santhakumaran S, Pan H, Modi N, on behalf of the Neonatal Data Analysis Unit and the Preterm Growth Investigator Group. Birth weight and longitudinal growth in infants born before 32 weeks’ gestation: a UK population study. Arch Dis Child Fetal Neonatal Ed 2014;99:F34-40
- Kuzma-O’Reilly B1, Duenas ML, Greecher C, Kimberlin L, Mujsce D, Miller D, Walker DJ. Evaluation, development, and implementation of potentially better practices in neonatal intensive care nutrition. Pediatr 2003;111:e461-70
- Peters O, Ryan S, Matthew L, Cheng, K. Randomised controlled trial of acetate in preterm neonates receiving parenteral nutrition. Arch Dis Child Fetal Neonatal Ed 1997;77:F12-F15
- Bauer J, Werner,C, Gerss. J. Metabolic rate analysis of healthy preterm and full- term infants during the first weeks of life. Am J Clin Nutr 2009;90:1517–24
- Koletzko B, Poindexter B, Uauy R. Nutritional care of preterm infants : scientific basis and practical guidelines. Basel: S. Karger AG; 2014.
- Koletzko B, Goulet O, Hunt J, Krohn K, Shamir R. Guidelines on Paediatric Parenteral Nutrition of the European Society of Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and the European Society for Clinical Nutrition and Metabolism (ESPEN), Supported by the European Society of Paediatric Research (ESPR). J Pediatr Gastroenterol Nutr 2005;41 Suppl 2:S1- 87.
- Senterre T, Rigo J. Reduction in postnatal cumulative nutritional deficit and improvement of growth in extremely preterm infants. Acta Paediatr 2012;101:e64- 70.
- Rochow N, Fusch G, Muhlinghaus A, Niesytto C, Straube S, Utzig N et al. A nutritional program to improve outcome of very low birth weight infants. Clin Nutr 2012;31:124-31.
- Johnson MJ, Wootton SA, Leaf AA, Jackson AA. Preterm birth and body composition at term equivalent age: a systematic review and meta-analysis. Pediatr 2012;130:e640-9.
- Uthaya S, Liu X, Babalis, D, Dore CJ, Warwick J, Bell J et al. Nutritional Evaluation and Optimisation in Neonates; a randomized double-blind controlled trial of amino acid regimen and intravenous lipid composition in preterm parenteral nutrition. Am J Clin Nutr e-pub ahead of print April 20, 2016 doi: 10.3945/ajcn.115.125138.
- Bottino M, Cowett RM, Sinclair JC. Interventions for treatment of neonatal hyperglycaemia in very low birthweight infants. Cochrane Database Syst Rev 2011;10:CD00745
- Stephens BE, Walden RV, Gargus RA, Tucker R, McKinley L, Mance M et al. First-week protein and energy intakes are associated with 18-month developmental outcomes in extremely low birth weight infants. Pediatr 2009;123: 1337–43
- Arsenault D, Brenn M, Kim S, Gura K, Compher C, Simpser E. ASPEN clinical guidelines: Hyperglycemia and hypoglycemia in the neonate receiving parenteral nutrition. J Parenter Enteral Nutr 2012;36:81-95.
- Lafeber HN, van de Lagemaat M, Rotteveel J, van Weissenbruch M. Timing of nutritional interventions in very-low-birth-weight infants: optimal neurodevelopment compared with the onset of the metabolic syndrome. Am J Clin Nutr 2013;98:556S-60S
- Cormack BE, Bloomfield FH. Increased protein intake decreases postnatal growth faltering in ELBW babies Arch Dis Child Fetal Neonatal Ed 2013;98:F399- 404.
- Mahaveer A, Grime C, Morgan C. Increased early protein intake is associated with a reduction in insulin-treated hyperglycaemia in very preterm infants. Nutr Clin Pract 2012;27:399-405.
- Te Braake FWJ. Van den Akker CHP. Riedijk MA, Goudoever JB. Parenteral amino acid and energy administration to premature infants in early life. Sem Fetal Neonatal Med 2007;12:11-18.
- Trevidi A, Sinn JKH. Early versus late administration of amino acids in preterm infants receiving parenteral nutrition (review).Cochrane Database of Systematic Reviews 2013: CD008771.
- Vlaardingerbroek H, Vermeulen MJ, Rook D, van den Akker CHP, Dorst K, Wattimena JL et al. Safety and efficacy of early parenteral lipid and high dose amino acid administration to very low birth weight infants J Pediatr 2013;163:638- 44.
- Burattini I, Bellagamba MP, Spagnoli C et al. Targeting 2.5 versus 4g/kg/day of amino acids for extremely low birth weight infants: randomised clinical trial. J Pediatr 2013;163:1278-82
- Moltu SJ, Strommen K, Blakstad EW Almaas AN, Westerberg AC, Brække K et al. Enhanced feeding in very-low-birth-weight infants may cause electrolyte disturbances and septicaemia- a randomised controlled trial. Clin Nutr 2012;32:207-12.
- Blanco CL, Gong AL, Green BK, Falck A, Schoolfield J, Liechty EA. Early changes in plasma amino acid concentrations during aggressive nutritional therapy in extremely low birth weight infants. J Pediatr 2011;158:543-8.
- Morgan C, Burgess L. High protein intake does not prevent low plasma levels of conditionally essential amino acids in very preterm infants receiving parenteral nutrition. J Parenter Enteral Nutr 2015 pii: 0148607115594009. [Epub ahead of print]
- Moyses HE, Johnson MJ, Leaf AA, Cornelius VR. Early parenteral nutrition and growth outcomes in preterm infants: a systematic review and meta-analysis. Am J Clin Nutr 2013;97:816-26
- Lee EJ, Simmer K, Gibson RA. Essential fatty acid deficiency in parenterally fed preterm infants. J Paediatr Child Health 1993;29:51-55.
- Salama GSA, Kaabneh MAF, Almasaeed MN, Alquran MIA. Intravenous IV lipids for preterm infants: a review. Clin Med Insights Pediatr 2015;9:25–36.
- Drenchpohl D, McConnell C, Gaffney S, Niehaus M, Macwan KS. Randomized trial of low birthweight infants receiving higher rates of infusion of intravenous IV fat emulsions during the first week of life. Pediatr 2008:122:743-51.
- Rayyan M, Devlieger H, Jochum F, Allegaert K. Short-term use of parenteral nutrition with a lipid emulsion containing a mixture of soybean oil, olive oil, medium-chain triglycerides, and fish oil. A randomized double-blind study in preterm infants. J Parenter Enteral Nutr 2012;36:81S–94S.
- Vlaardingerbroek H, Veldhorst M, Spronk S et al. Parenteral lipid administration to very-low-birth-weight infants—early introduction of lipids and use of new lipid emulsions: a systematic review and meta-analysis. Am J Clin Nutr 2012;96:255– 68.
- Haumont D , Deckelbaum RJ, Richelle M, Dahlan W, Coussaert E, Bihain BE, Carpentier YA. Plasma lipid and plasma lipoprotein concentrations in low birth weight infants given parenteral nutrition with twenty or ten percent lipid emulsion. J Pediatr. 1989 Nov;115(5 Pt 1):787-93.
- van Kempen AA, van der Crabben SN, Ackermans MT, et al. Stimulation of gluconeogenesis by intravenous IV lipids in preterm infants: Response depends on fatty acid profile. Am J Physiol Endocrinol Metab. 2006;290:e723–e730.
- Bonsante F, Iacobelli S, Latorre G, Rigo J, De Felices C, Robillard PY, Gouyon B. Initial Amino Acid Intake Influences Phosphorus and Calcium Homeostasis in Preterm Infants – It Is Time to Change the Composition of the Early Parenteral Nutrition. PLoS ONE 2013 ;8:e72880.doi:10.1371/journal.pone.00728801.
- Parenteral Nutrition in a Neonatal Intensive Care Unit: Galenic Stability of Four All-in-one Admixtures, E.Bourgier, S. Poullan-Termeu, Eur J Hosp Pharm 2015:22:285-290.
- Greene HL, Hambidge KM, Schanler R, Tsang RC. Guidelines for the use of vitamins, trace elements, calcium, magnesium, and phosphorus in infants and children receiving total parenteral nutrition: report of the Subcommittee on Pediatric Parenteral Nutrient Requirements from the Committee on Clinical Practice Issues of the American Society for Clinical Nutrition. Am J Clin Nutr 1988;48:1324-42.
- Berkow SE, Spear ML, Stahl GE, Gutman A, Polin RA, Pereira GR, Olivecrona T, Hamosh P, Hamosh M. Total parenteral nutrition with intralipid in pr Infants Receiving TPN with Heparin: Effect c Lipolytic Enzymes, Lipids, and Glucose. J Pediatr Gastroenterol Nutr 1987;6:581-588.
- Zingg W, Tomaske M, Martin M. Risk of parenteral nutrition in neonates – an overview. Nutrients 2012;4:1490-1503.
- http://www.bapm.org/publications/documents/guidelines/Use%20of%20Central% 20Venous%20Catheters%20in%20neonates.pdf. Accessed 6.12.15
- Reece A, Ubhi T, Craig AR, Newell SJ. Positioning long lines: contrast versus plain radiography. Arch Dis Child Fetal Neonatal Ed 2001;84:F129-130.
- Rorke JM, Ramesthu J. Ch 31 Percutaneous central venous catheterisation. MacDonald MG, Ramasathu J (Eds) 2002; Procedures in Neonatology 3 rd Ed Lippincott Williams and Wilkins Philiadelphia.
- Mirtallo J, Canada T, Johnson D, Kumpf V, Peterson c, Sacks G, Seres D, Guenter P. Safe Practices for Parenteral Nutrition. J Parenter Enteral Nutr 2004;28:S39-S70.
- Cies JJ, Moore WS. Neonatal and Pediatric peripheral parenteral nutrition: What is a safe osmolarity? Nutr Clin Pract 2014;29:118-124.
- Dugan S, Le J, Jew RK. Maximum tolerated osmolarity for peripheral administration of peripheral parenteral nutrition in pediatric patients. J Parenter Enteral Nutr 2014;38(7):847-851.
- Pineault M, Chessex P, Piedboeuf B, Bisaillon S. Beneficial effect of coinfusing a lipid emulsion on venous patency. J Parenter Enteral Nutr 1989;13(6):637-640.
- Foster JP, Richards R, Showell MG, Jones LJ. Intravenous IV in-line filters for preventing morbidity and mortality in neonates. Cochrane Database of Systematic Reviews 2015, Issue 8. Art. No.: CD005248. DOI: 10.1002/14651858.CD005248.pub3.
- Bethune K, Allwood M, Grainger C, Wormleighton C; British Pharmaceutical Nutrition Group Working Party. Use of filters during the preparation and administration of parenteral nutrition: position paper and guidelines prepared by a British pharmaceutical nutrition group working party. Nutrition 2001;17:403-8.
- Balegar VK, Azeem MI, Spence K, Badawi N: Extending total parenteral nutrition hang time in the neonatal intensive care unit: is it safe and cost effective? J Paediatr Child Health 2013;49:E57–E61.
- Fox M, Molesky M, Van Aerde JE, Muttitt S: Changing parenteral nutrition administration sets every 24 h versus every 48 h in newborn infants. Cana J Gastroenterol 1999;13:147–151.
- Matlow AG, Kitai I, Kirpalani H, Chapman NH, Corey M, Perlman M et al. A randomized trail of 72- versus 24-hour intravenous IV tubing set changes in newborns receiving IV lipid therapy. Inf Control Hosp Epidem 1999;20:487-493.
- Silvers KM, Sluis KB, Darlow BA, McGill F, Stocker R, Winterbourn CC. Limiting light-induced lipid peroxidation and vitamin loss in infant parenteral nutrition by adding multivitamin preparations to Intralipid. Acta Pediatr 2001;90:242-9.
- Khashu M, Harrison A, Lalari V, Lavoie J-C, Chessex P. Impact of shielding parenteral nutrition from light on routine monitoring of blood glucose and triglyceride levels in preterm neonates. Arch Dis Child Fetal Neonatal Ed 2009;94:F111-115.
- Laborie S, Denis A, Dassieu G, Bedu A, Tourneux P, Pinquier D et al. Shielding parenteral nutrition solutions from light: a randomised, controlled study. J Parenter Enteral Nutr 2015;39:729-37.
- Peirovifar A, Gharehbaghi MM, Abdulmohammad-zadeh H, Sadegi GH, Jouyban A. Serum selenium levels of the very low birth weight premature newborn infants with bronchopulmonary dysplasia. Journal of Trace Elements in Medicine and Biology 2013;27:317-321.
- Miller M, Vaidya R, Rastogi D, Bhutada A, Rastogi S. From parenteral to enteral nutrition: a nutrition-based approach for evaluating postnatal growth failure in preterm infants. J Parenter Enteral Nutr 2014;38:489-97.
- Lapillonne A, Fellous L, Mokthari M, Kermorvant-Duchemin E. Parenteral nutrition objectives for very low birth weight infants: results of a national survey. J Pediatr Gastroenterol Nutr. 2009;48:618-26.
Naveed RivasParticipant1- who will do the counseling?
Pharmacists and pharmacy interns under supervision of pharmacist even your manager, assistant manager and supervisor can also go for counseling. usually we have one pharmacist on clinical rotation from every area so he or she is doing discharge counseling but he is not 24/7 and patients are getting discharge 24/7 to cope up with this situation we have separate discharge pharmacy (take home pharmacy) their staff is going for counseling. we have designed education material for patient education in form of booklets and pamphlets in case pharmacist or intern is not available for discharge counseling that is a rare scenario.
2- whom will be eligible to be counseled? Any criteria?
When we started discharge counseling ( five years back) at that time our target group was cardiac patients especially patients on warfarin and other anticoagulants as we have started anticoagulation clinics also, transplant patients, pediatric patients, oncology patients and patients getting discharge on pharmacy compounding but now it is extended to all discharge patients.
3- when the physician write a discharge medications, do the clinical pharmacist review those medications before being dispensed by pharmacist.
In akuh (Pakistan) we don’t have two terms in practice i.e. pharmacist and clinical pharmacist as we have six years Pharm-D graduates (5 years of Pharm D and one year internship) so our pharmacists are clinical pharmacists. our process flow is physician enter discharge order in CPOE then one pharmacist process the order after that technician fill the order as we don’t have automated prescription filling system, if you have automated prescription filling then it will add another feather in your cap, then second pharmacist review that order. our goal is to dispense the discharge medication within 45 mins.
-
AuthorPosts

